Cells, Free Full-Text
Por um escritor misterioso
Descrição
Community-acquired pneumonia remains a major contributor to global communicable disease-mediated mortality. Neutrophils play a leading role in trying to contain bacterial lung infection, but they also drive detrimental pulmonary inflammation, when dysregulated. Here we aimed at understanding the role of microRNA-223 in orchestrating pulmonary inflammation during pneumococcal pneumonia. Serum microRNA-223 was measured in patients with pneumococcal pneumonia and in healthy subjects. Pulmonary inflammation in wild-type and microRNA-223-knockout mice was assessed in terms of disease course, histopathology, cellular recruitment and evaluation of inflammatory protein and gene signatures following pneumococcal infection. Low levels of serum microRNA-223 correlated with increased disease severity in pneumococcal pneumonia patients. Prolonged neutrophilic influx into the lungs and alveolar spaces was detected in pneumococci-infected microRNA-223-knockout mice, possibly accounting for aggravated histopathology and acute lung injury. Expression of microRNA-223 in wild-type mice was induced by pneumococcal infection in a time-dependent manner in whole lungs and lung neutrophils. Single-cell transcriptome analyses of murine lungs revealed a unique profile of antimicrobial and cellular maturation genes that are dysregulated in neutrophils lacking microRNA-223. Taken together, low levels of microRNA-223 in human pneumonia patient serum were associated with increased disease severity, whilst its absence provoked dysregulation of the neutrophil transcriptome in murine pneumococcal pneumonia.

The cell-free system: A new apparatus for affordable, sensitive, and portable healthcare - ScienceDirect
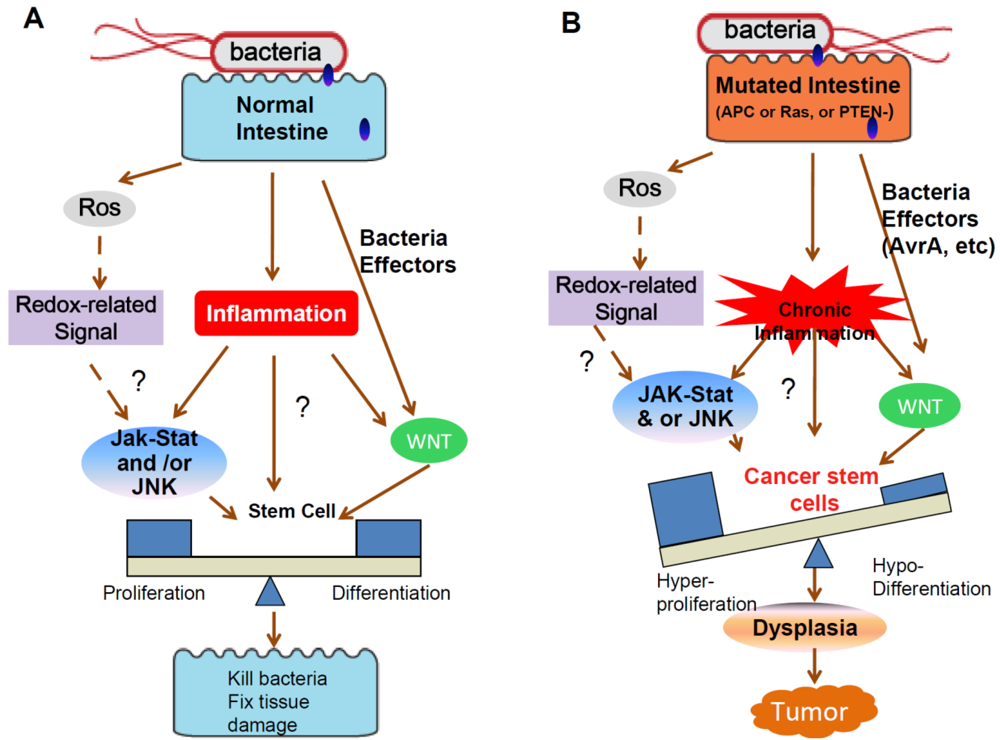
Cancers, Free Full-Text
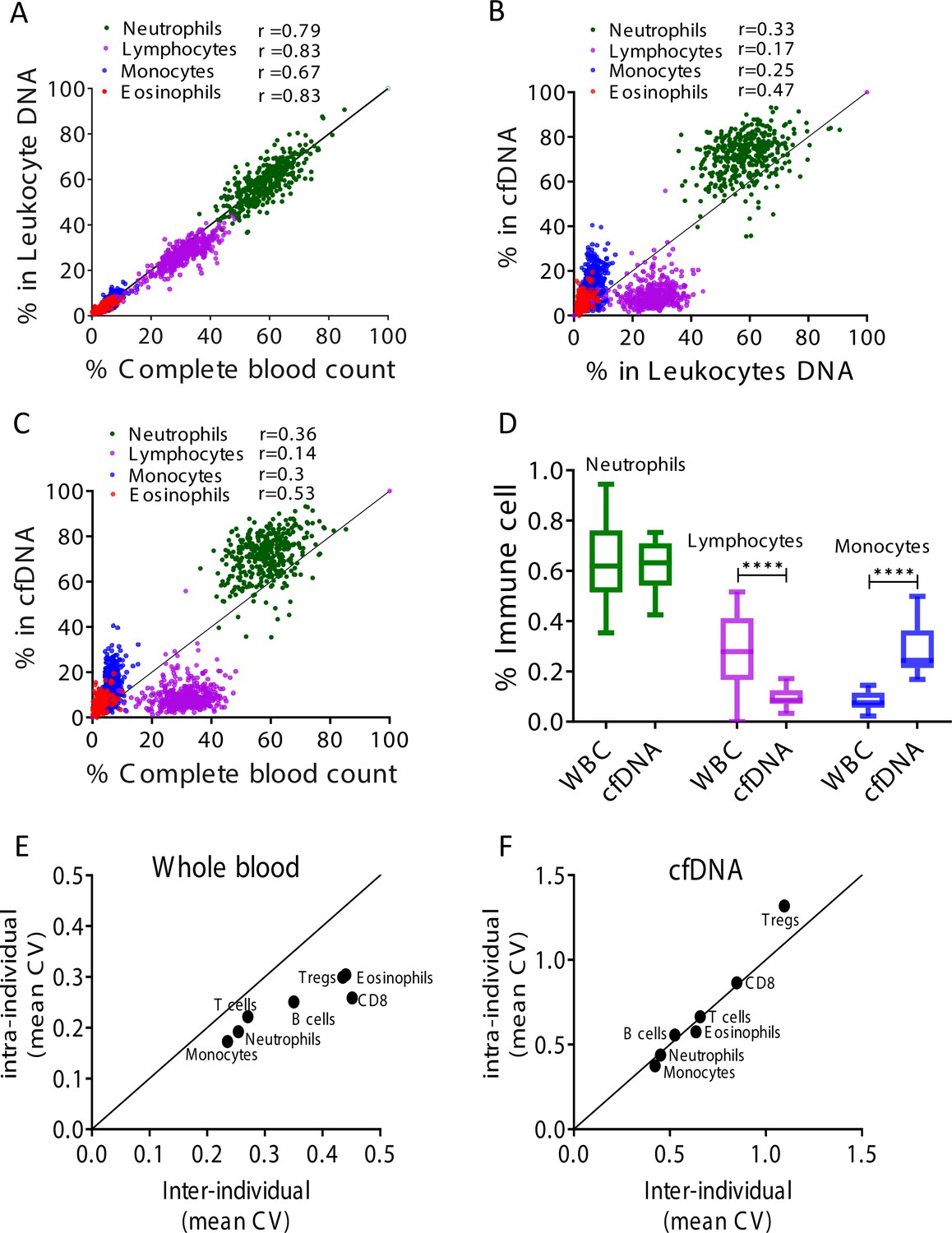
Remote immune processes revealed by immune-derived circulating cell-free DNA
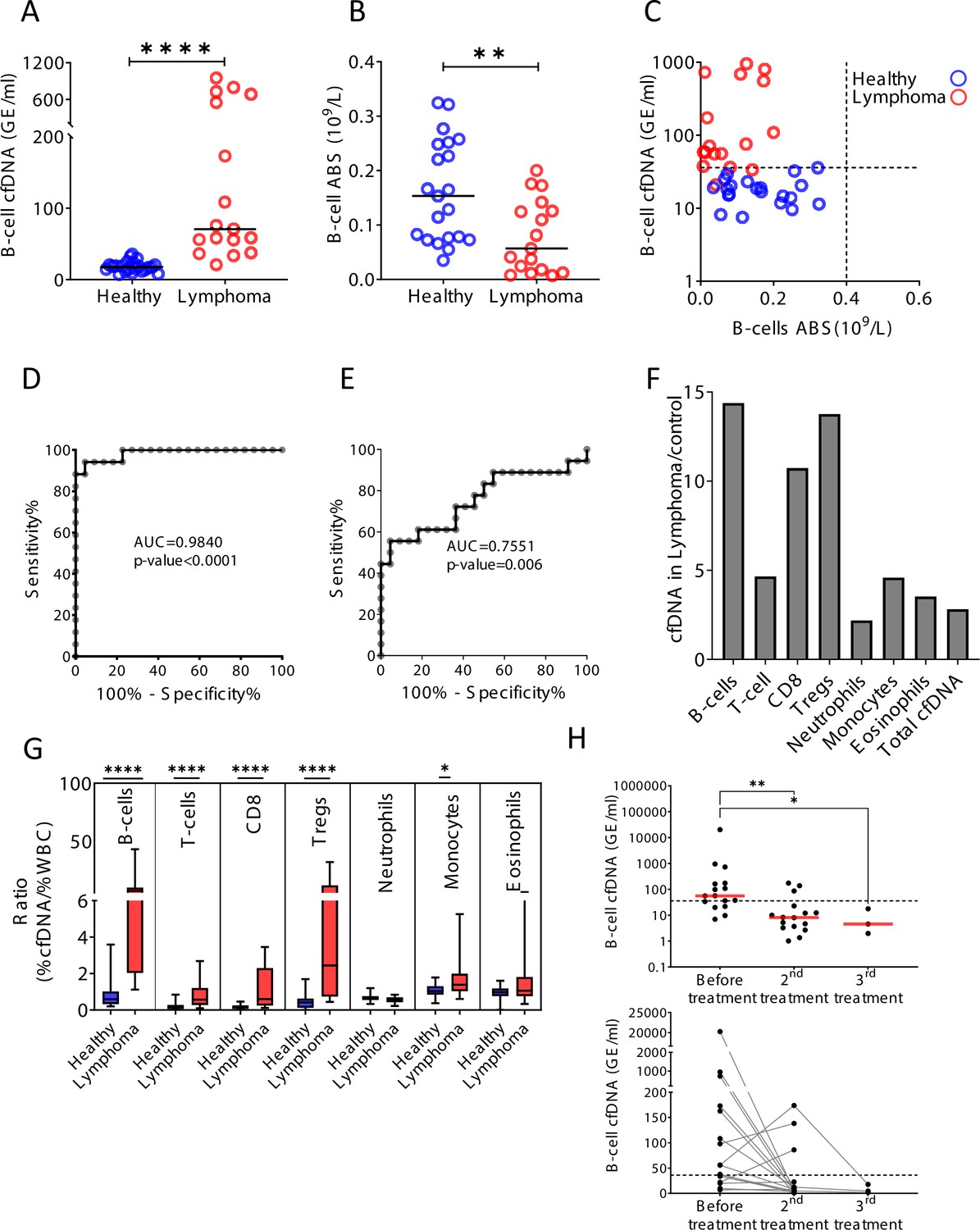
Remote immune processes revealed by immune-derived circulating cell-free DNA

Nucleic acid biomarkers of immune response and cell and tissue damage in children with COVID-19 and MIS-C - ScienceDirect

Cells, Free Full-Text
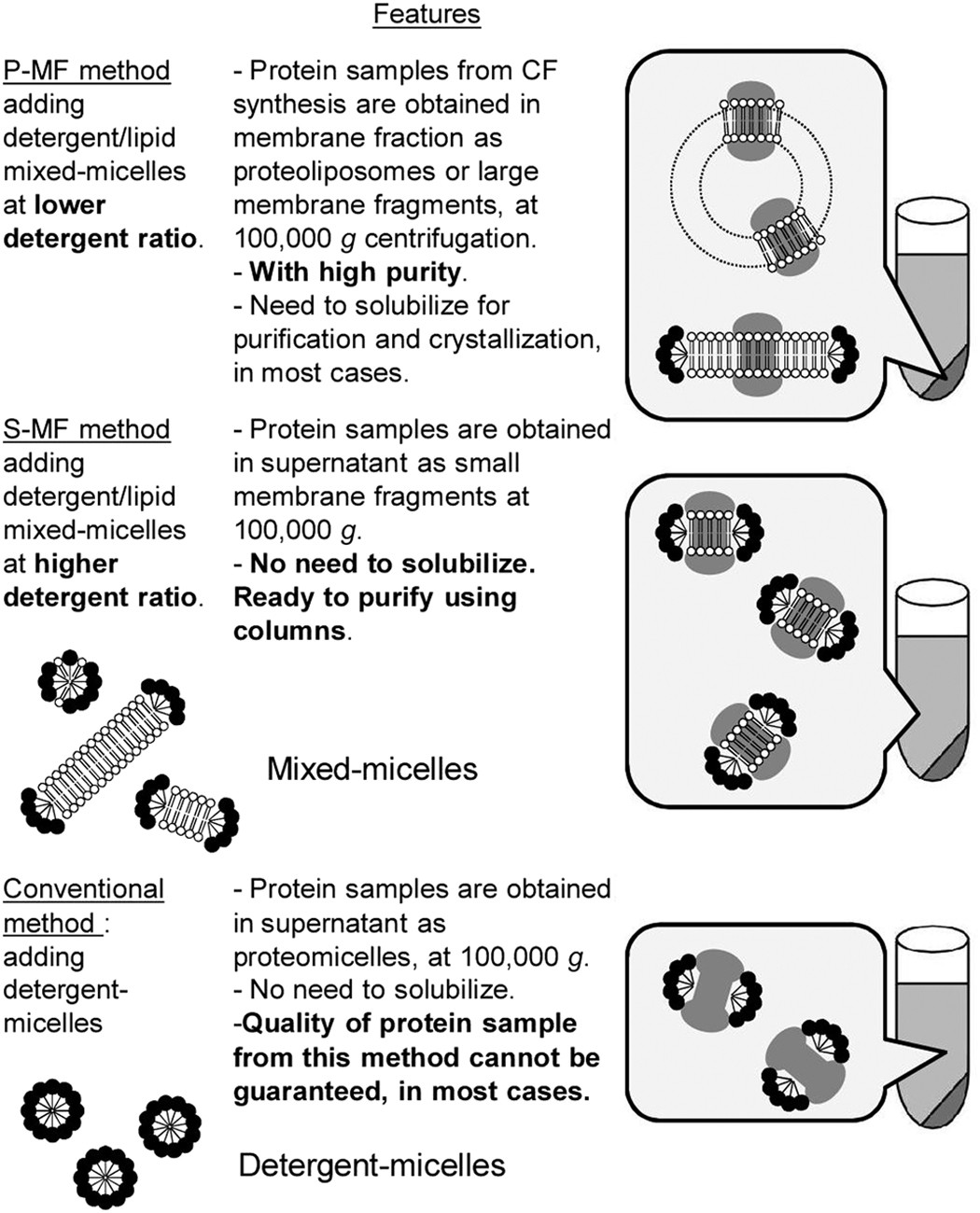
Cell-free methods to produce structurally intact mammalian membrane proteins
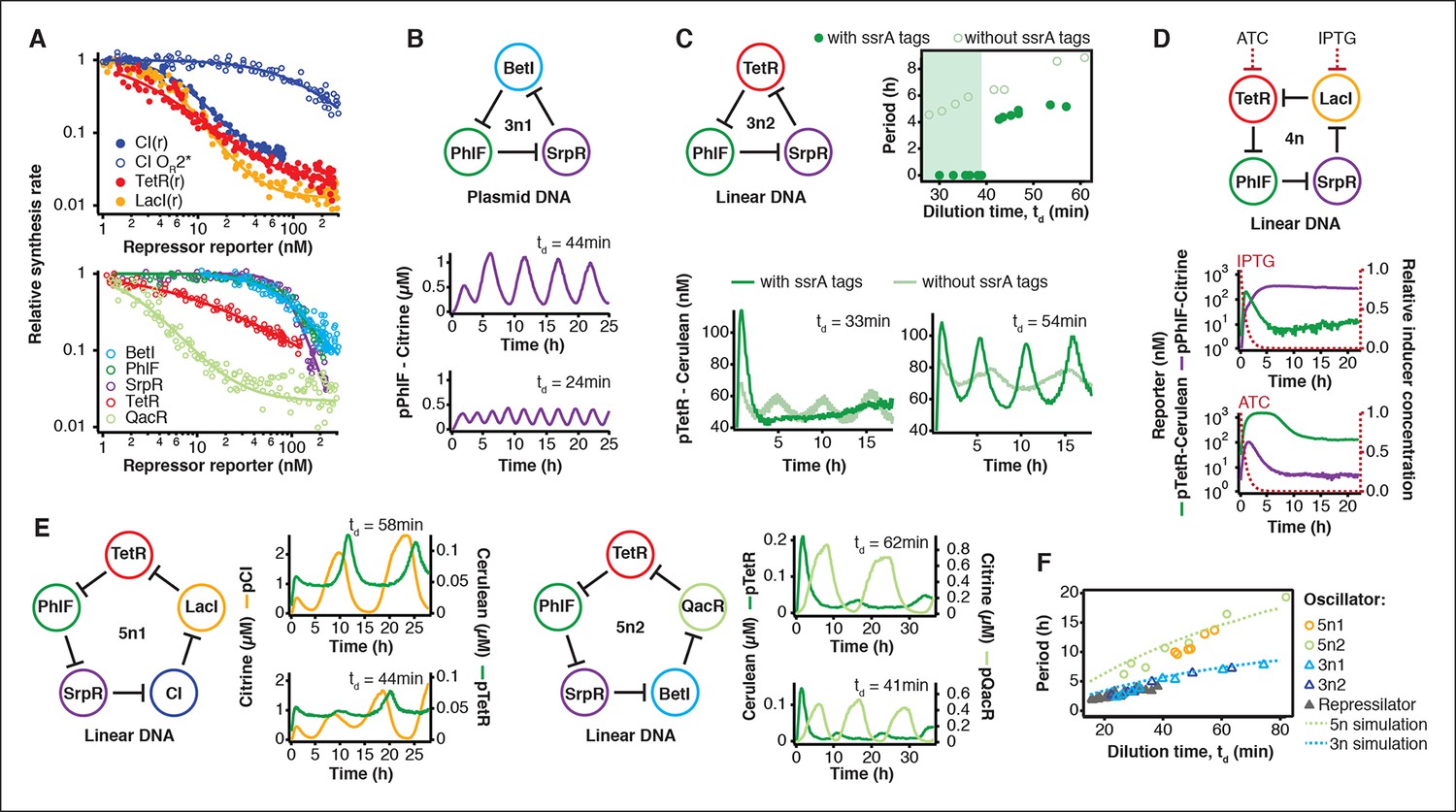
Rapid cell-free forward engineering of novel genetic ring oscillators
Cells, Free Full-Text
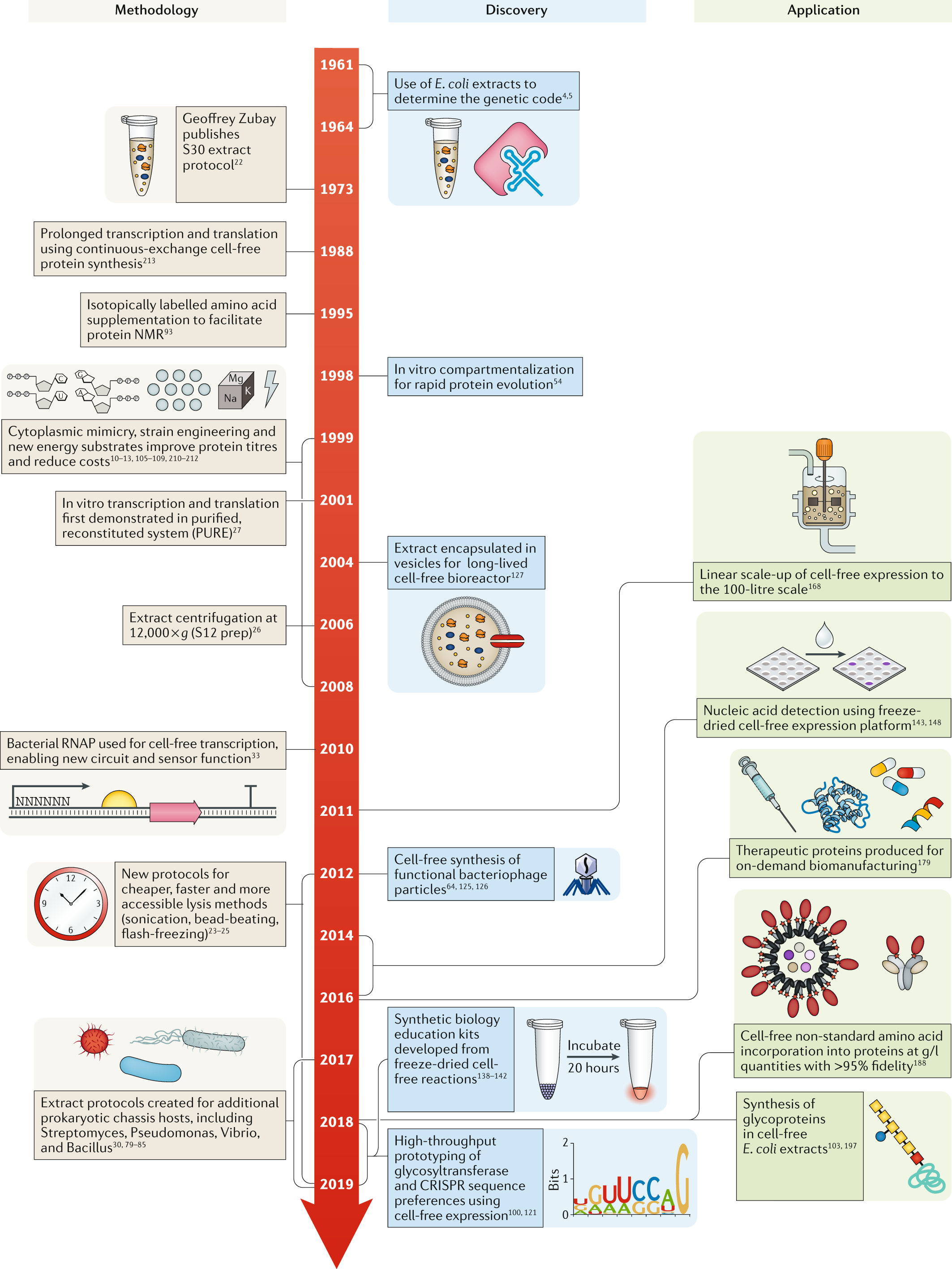
Cell-free gene expression: an expanded repertoire of applications

Cells, Free Full-Text
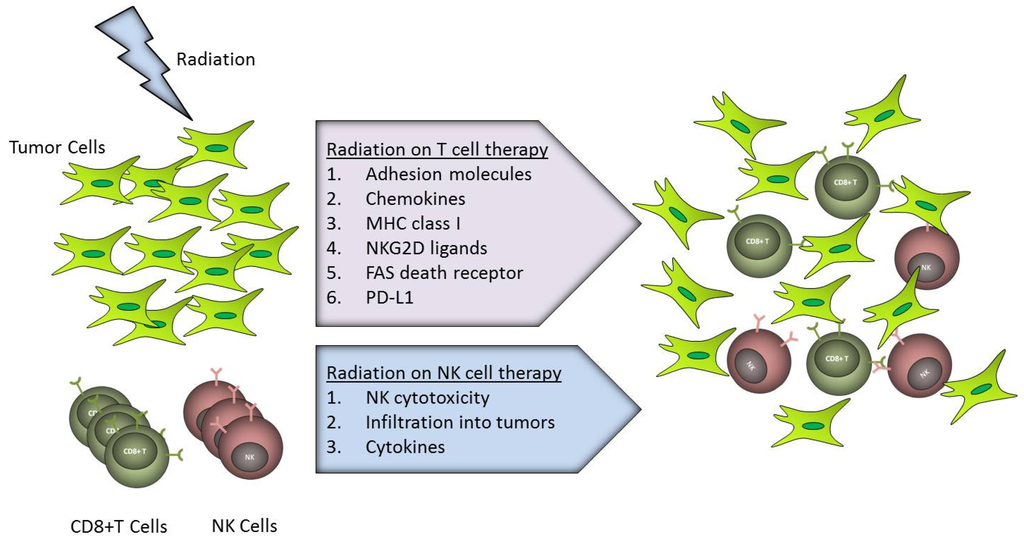
IJMS, Free Full-Text
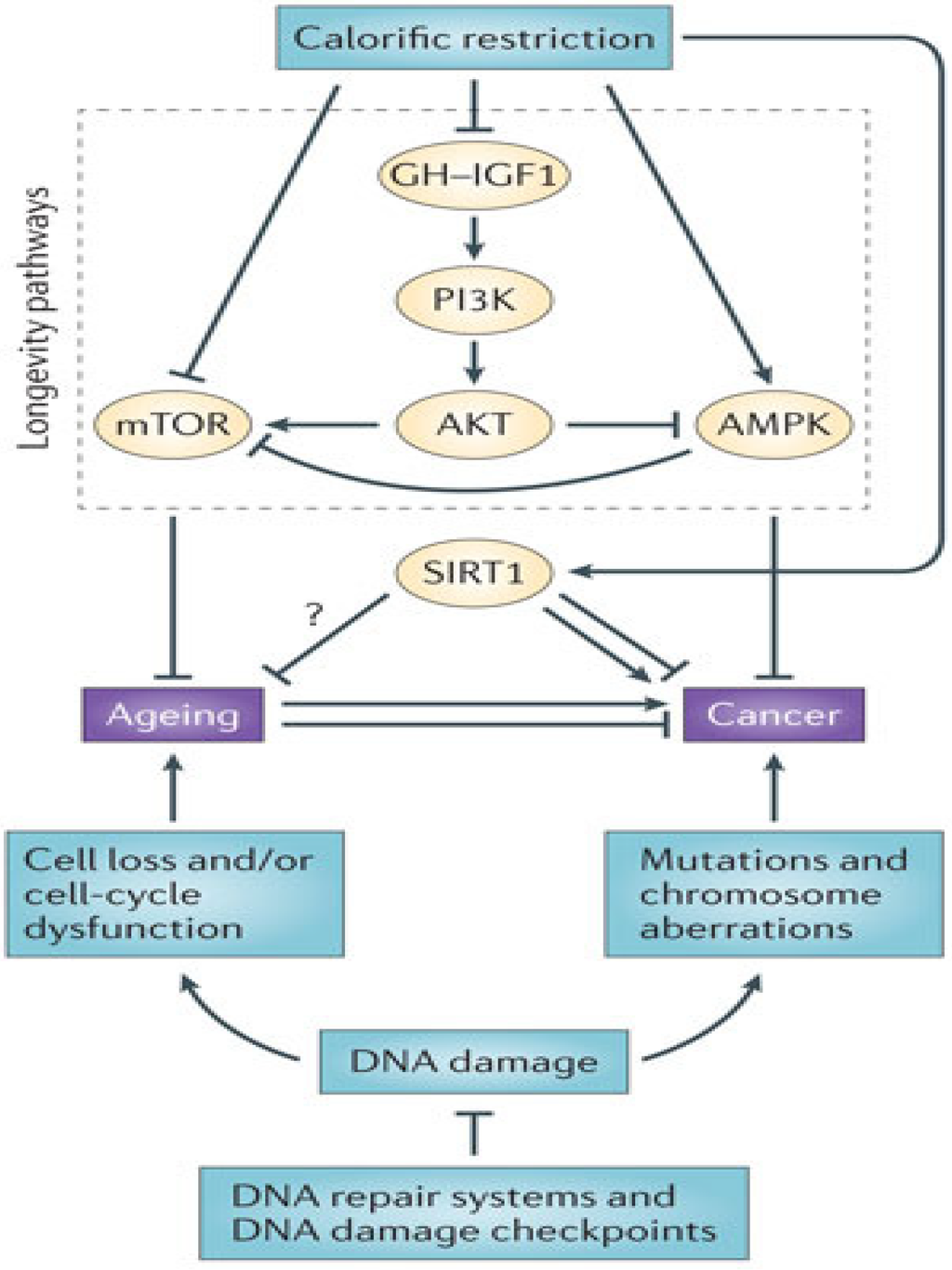
JCM, Free Full-Text
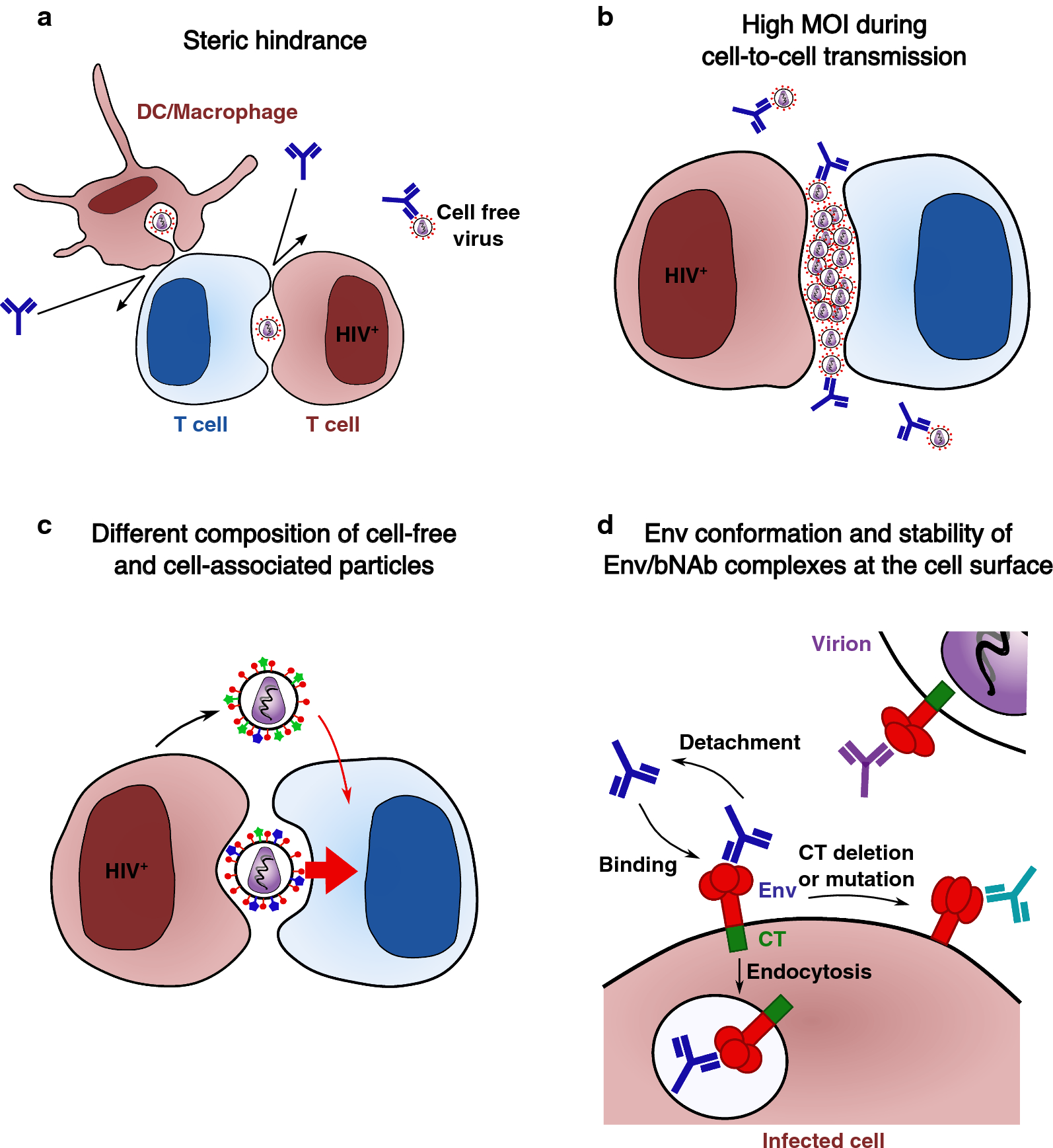
HIV-1 cell-to-cell transmission and broadly neutralizing antibodies, Retrovirology
de
por adulto (o preço varia de acordo com o tamanho do grupo)






