hydrogen orbital wavefunction
Por um escritor misterioso
Descrição

Hydrogen Atom Wave Function Electron Orbital (4,3,0) Poster for Sale by cbeuw

Hydrogen orbital wavefunction : r/desmos

For `3s` orbital of hydrogen atom, the normalised wave function is `Psi_(3s)=(1)/((81)

The angular components of the wave function of one of

Solved Given the wave function of the 1s orbital of the
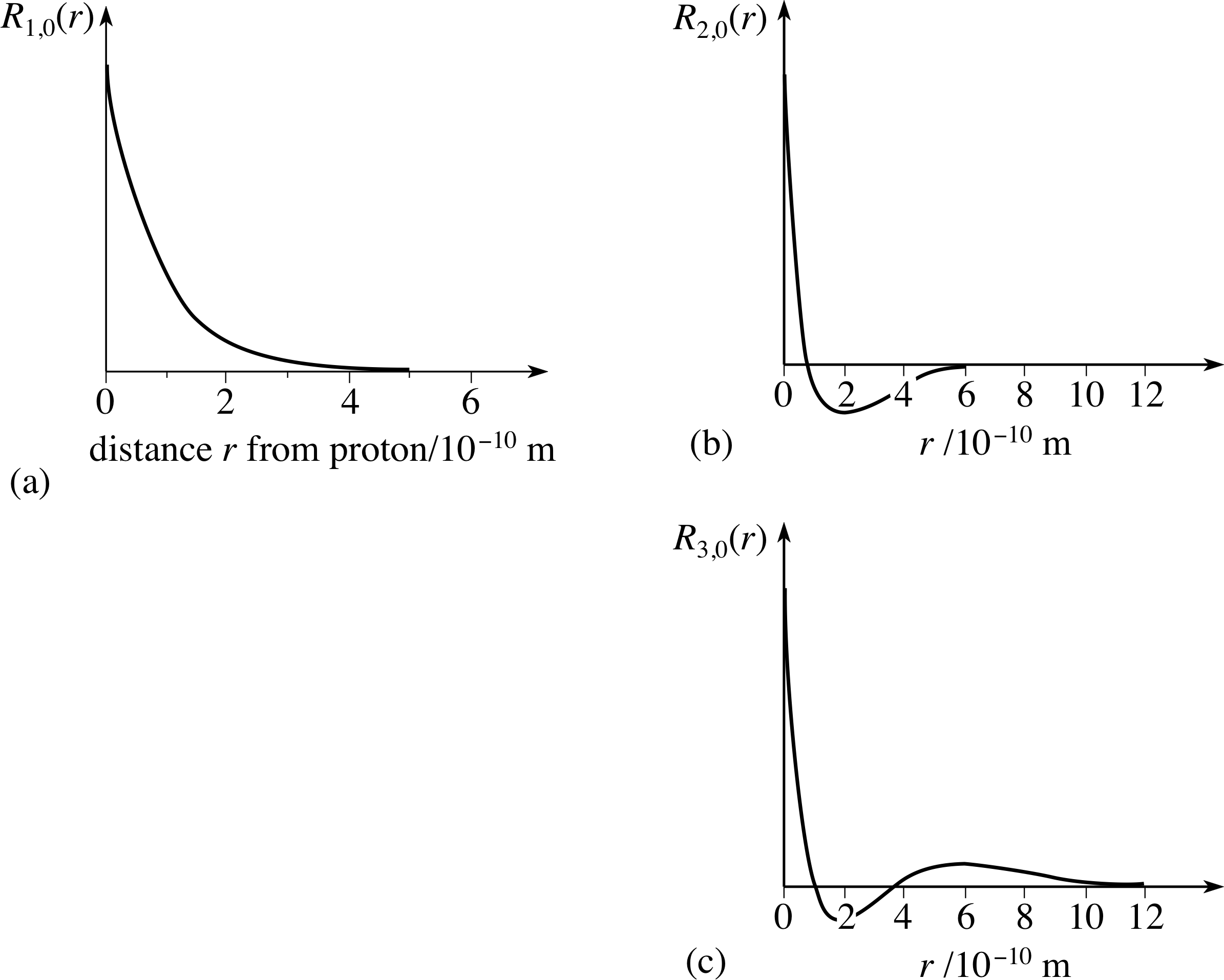
PPLATO, FLAP
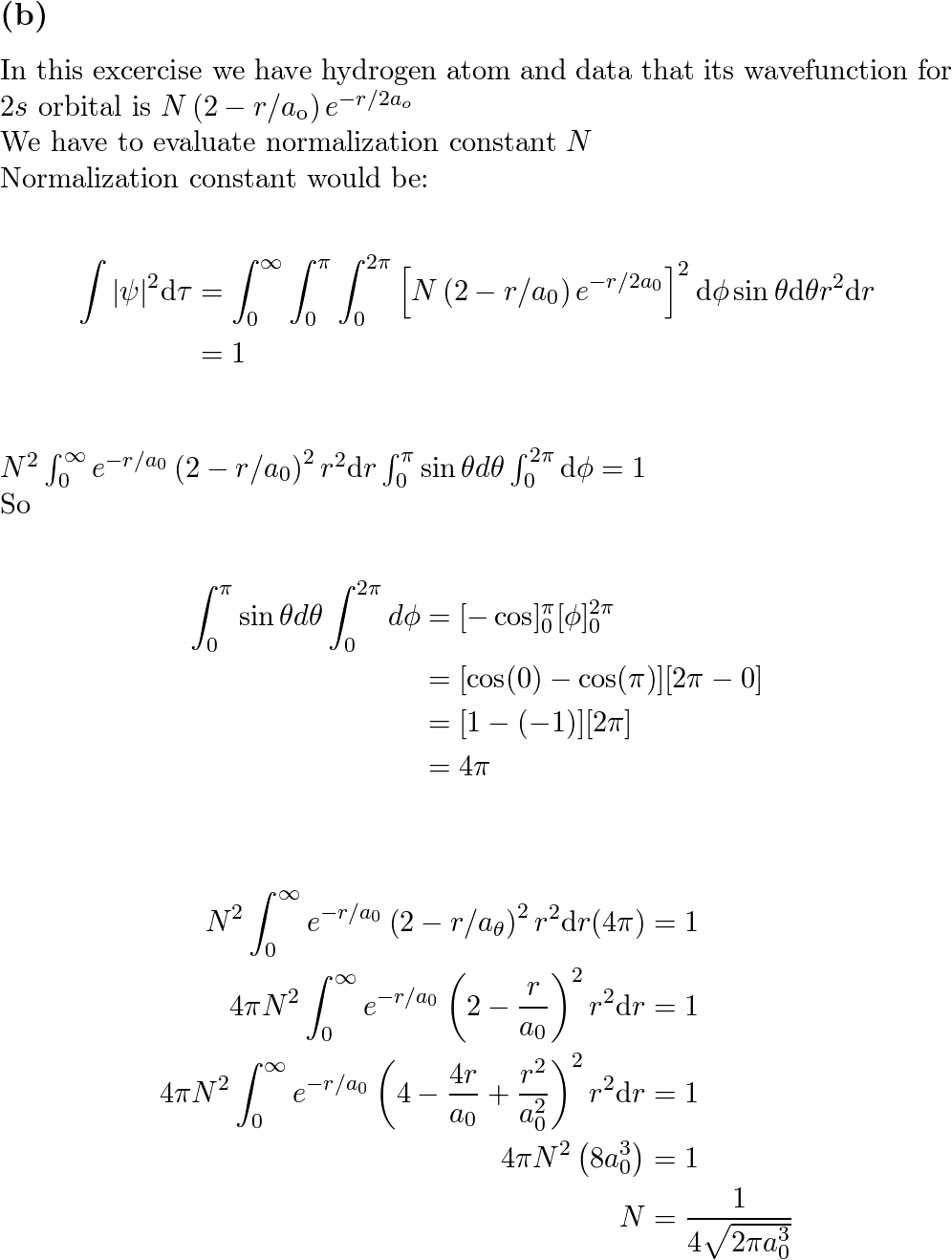
a) The wavefunction for the ground state of a hydrogen atom

The average distance of a 1s hydrogen electron, scenic-science + thematic-mathematics
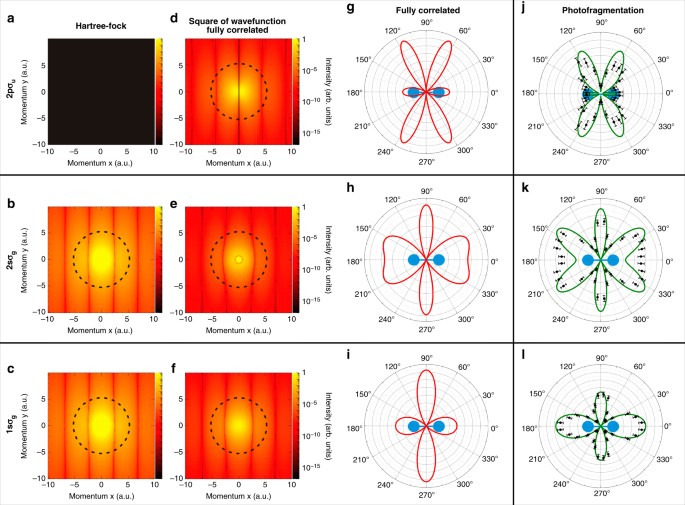
Imaging the square of the correlated two-electron wave function of a hydrogen molecule
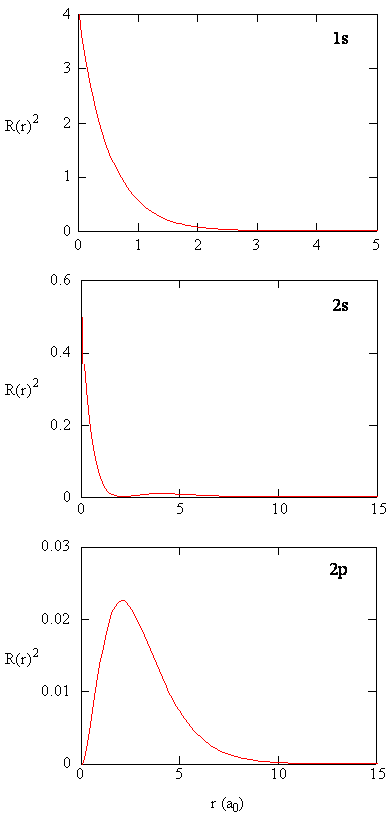
11.10: The Schrödinger Wave Equation for the Hydrogen Atom - Chemistry LibreTexts

Visualization of the hydrogen atom wave functions
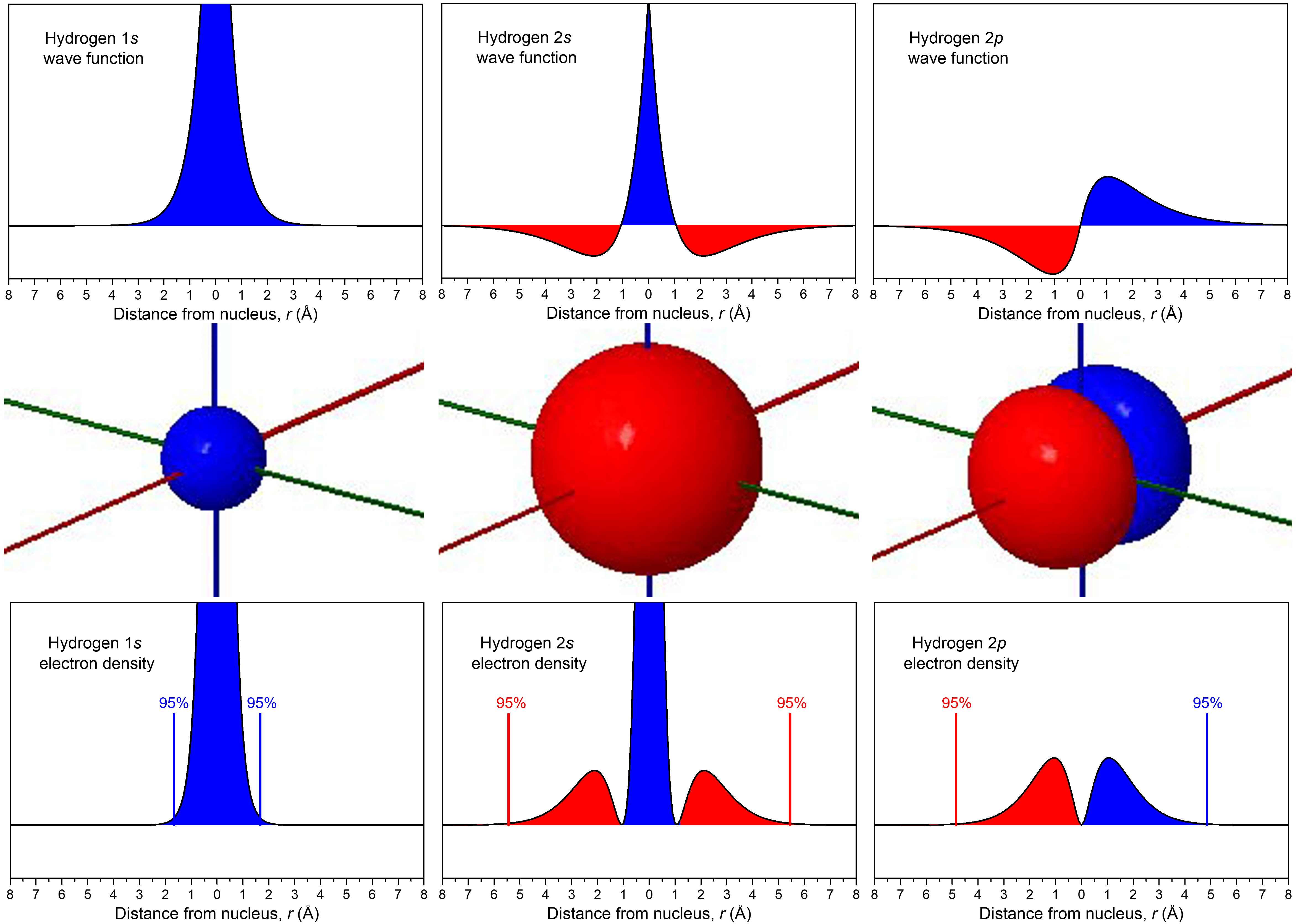
D3.1 Atomic Orbitals and Quantum Numbers – Chemistry 109 Fall 2021


Hydrogen atom wavefunctions

The wavefunction Psi _{n}, l, m_c is a mathematical whose value depends upon spherical polar coordinates (r, theta , phi) of the electron and charactrised by the quantum number n, l and
de
por adulto (o preço varia de acordo com o tamanho do grupo)






